Testing will proceed without the form however, results will be held under the information is received.įor serial monitoring, it is recommended to perform specimen collection at the same time of day, if possible. TCR V beta Spectratyping Assay Patient Information (T719) is required. Ordering physician's name and phone number are required. 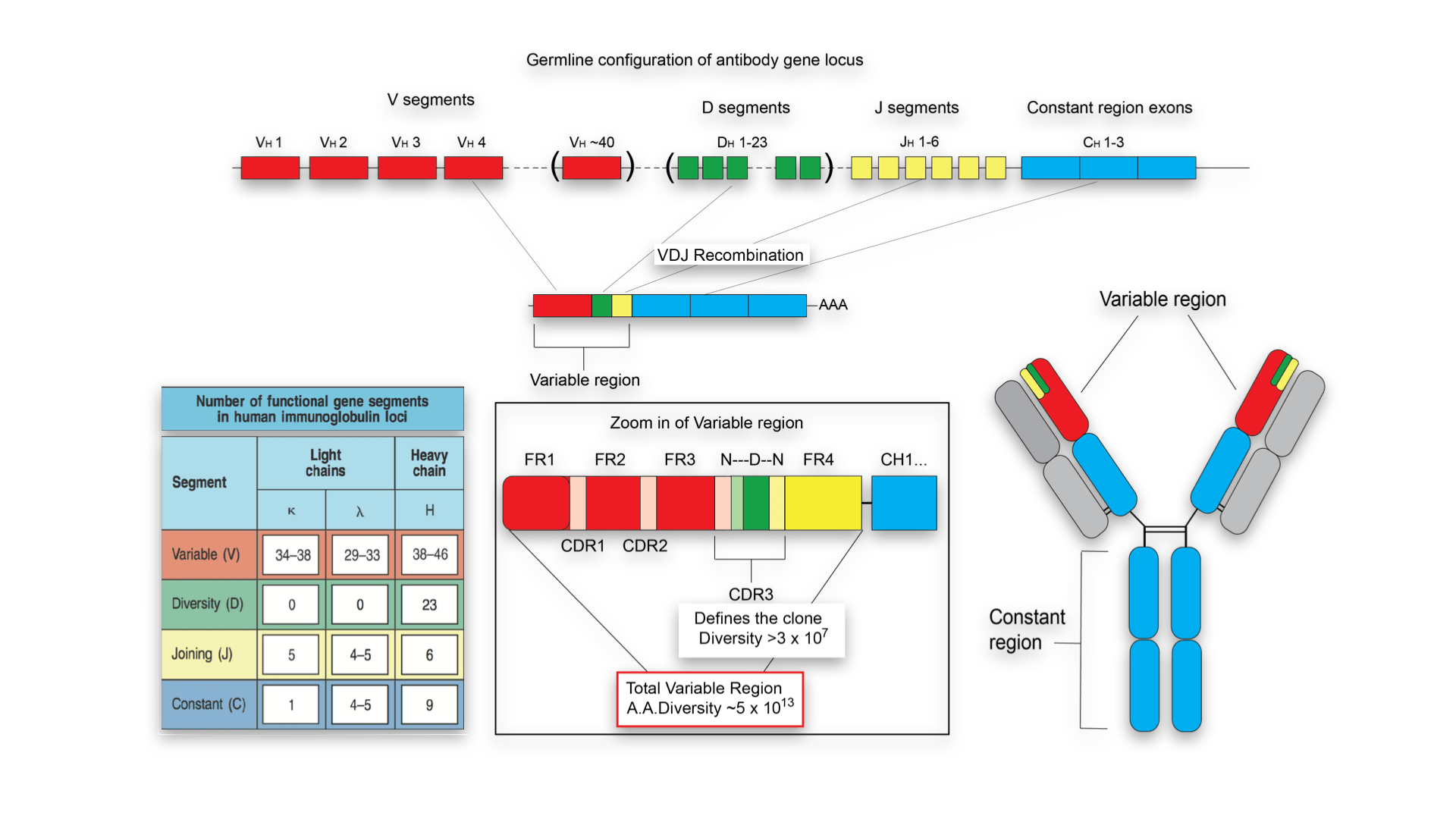
Samples arriving over the weekend and/or on observed holidays may be canceled. It is recommended that specimens arrive within 24 hours of collection. Collect and package specimen as close to shipping time as possible. Specimens must be received in the laboratory on weekdays and by 4 p.m. TCP / T-Cell Subsets, Naive, Memory, and Activated, Blood CD4RT / CD4 T-Cell Recent Thymic Emigrants, Blood TRECS / T-Cell Receptor Excision Circles (TREC) Analysis, Blood 
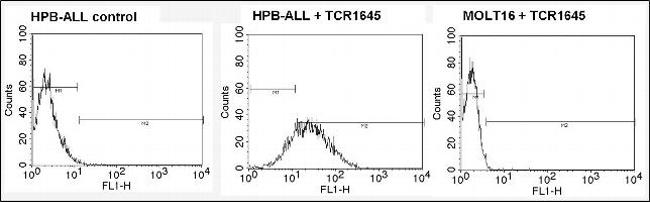
1994Īssessment of T-cell receptor repertoire by flow cytometry using the Beta Mark TCR Vβ repertoire kit and Kaluza analysis softwareĪuthor: Brice Ezzouaouy, Caroline Scifo | Beckman Coulter Immunotech S.A.Mayo Clinic Laboratory Director/Consultant approval is required prior to ordering this test in patients greater than 40 years of age.Īdditional tests that could be ordered in conjunction with this test include: The extent of the human germline T-cell receptor Vβ gene segment repertoire. Kit antibody composition and associated Vbeta according to Wei, et al. Since the reagent in vial A is used as to prepare this control, the kit contains 50 tests of reagent A instead of 25. The IOTest Beta Mark kit includes a control for PMT setting and fluorescence compensation adjustments. The test is compatible with different methods of red blood cell lysis and can be run on Beckman Coulter as well as BD Biosciences cytometers. The kit can be used in a multi parametric environment employing at least three-color flow cytometric analysis. A third color T-cell marker (preferentially as PC5-conjugate not included) is used to gate the specific population. The staining protocol includes a one-step procedure with directly conjugated antibody mixes (test volume of 20 μL) and a wash step after staining. The test is intended for use on whole blood samples. The kit is composed of 8 vials containing mixtures of conjugated TCR Vβ antibodies corresponding to 24 different specificities (about 70% coverage of normal human TCR Vβ repertoire). Moreover, this kit makes it possible to study the repertoire on T cell subsets, using additional T-cell markers, such as CD3, CD4, and CD8, conjugated to a third fluorophore. This kit simplifies the TCR Vβ repertoire analysis by reducing the number of tubes to analyze (8 instead of 24) and consequently the time required for obtaining the results. A first mAb is FITC-conjugated, a second one is PE-conjugated and a third one is a carefully balanced mixture of a PE- and a FITC-conjugated form. Taking advantage of the fact that Vβ specificities may be grouped into mutually exclusive combinations, the detection of 3 Vβ expressions in the same tube is possible with the use of an innovative staining strategy combining three monoclonal antibodies (mAb) with only two fluorophores. The IOTest Beta Mark Kit is a multi parametric analysis tool designed for quantitative determination of the TCR Vβ repertoire of human T lymphocytes by flow cytometry. Liquid Handling and Scheduling Software.CytExpert Software for the CytoFLEX Platform.Plate Loader Options for the CytoFLEX Platform.CytoFLEX Violet-Blue-Red Series Upgrades.Labware for Liquid Handling Instruments.Biomek NGeniuS Reaction Vessel, 24 Well, 64/Box.Lysing, Fixative, and Permeabilizating Reagents.RESOURCE Contract Manufacturing Services.HIAC 9703+ Pharmaceutical Particle Counter.Biomek NGeniuS Next Generation Library Prep System.Cell Counters, Sizers and Media Analyzers.Air Particle Counters for Cleanroom and Environmental Monitoring.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
